
QUANTUM TELEPORTATION: THE THEORY

QUANTUM
TELEPORTATION: THE
THEORY
Quantum Teleportation is a technique of communication that takes advantage of some unique aspects of the Quantum Mechanics.
The Quantum Mechanics is a physical theory that describes the behavior of the electromagnetic radiation, the matter and their interactions, particularly with regard to the phenomena typical of the length or energy scales of the atomic and subatomic particles.
The Quantum Mechanics developed in the first half of the twentieth century, due to the inconsistency of the Classical Mechanics and its inability to represent the experimental reality, with particular reference to the light and the electron.
The name “Quantum Mechanics”, was introduced by Max Planck in the early twentieth century; it is based on the fact that quantities such as energy or angular momentum of some physical systems can change in a discrete manner, namely assuming only certain values, named “quanta”.
The basic characteristic that distinguishes the Quantum Mechanics from the Classical Mechanics is that in the Quantum Mechanics the electromagnetic radiation and the matter are both described as a wave phenomenon and, at the same time, as particles, in contrast to the Classical Mechanics, where, for example, the light is described only as a wave and the electron only as a particle. This unexpected and non intuitive property called “wave-particle duality” is the main reason for the failure of all classical theories developed until the nineteenth century.
Some experimental phenomena, in which the Classical Physics fails, are listed below:
Black body radiation
Photoelectric Effect
Atomic spectral lines
Wave properties of the electrons
Quantum Mechanics provides solutions with:
Planck's theory of the radiation of a black body:
Max Planck found that the energy of the radiation emitted or absorbed by a black body is not emitted and absorbed continuously but in discrete quantities called “quanta”. The main concept of his theory was based on the fact that each elementary oscillator (the electrons within the atom) could exchange energy with the environment only in packets of E = hʋ, where h = 6,63×10-34 J·s = 6,63x10-27 erg·s is the Planck's constant and ʋ is the frequency of the oscillator.
Einstein's explanation of the photoelectric effect:
Albert Einstein was the first to recognize that the energy quantization of the emitted or absorbed radiation is a general property of the electromagnetic radiation, thinking of it as a set of photons with energy E = hʋ.
Bohr's model of the hydrogen atom:
Niels Bohr applied the ideas of Einstein, about energy quantization, at the energy of an atom. He proposed a model of the hydrogen atom which was a spectacular success in the calculation of the wavelengths of the radiation emitted by the hydrogen atom.
Wavelength of de Broglie:
Louis de Broglie was the first to assume the wave nature of matter.
WAVE – PARTICLE DUALITY
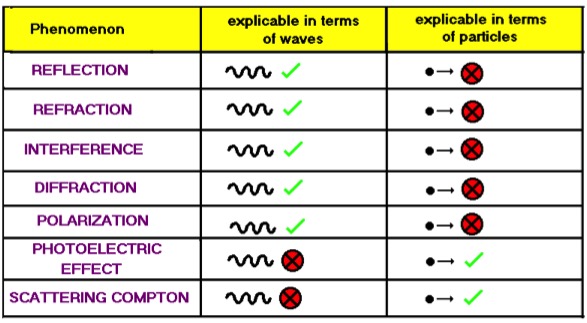
WAVE AND PARTICLE NATURE OF THE LIGHT: the light consists of particles or waves depending on the type of observed phenomena.
The most common luminous phenomena observed, such as reflection, refraction, interference and diffraction can be explained as wave phenomena. However, the light that usually we imagine as a wave shows also particle properties when it interacts with the matter, as demonstrated by the photoelectric effect and Compton scattering.
Schematic summary
WAVE NATURE OF THE MATTER: the wave-particle duality is also valid for the electrons and matter in general. The electrons, which we usually think as particles, have also wave properties of interference and diffraction.
Louis de Broglie, in 1924, stated that all the particles had wave properties. He affirmed that the wavelength associated with the wave of matter was inversely proportional to the mass “m” of the particle and to its velocity “v”, so that:
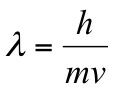
where “h” is Planck's constant
The product of mass and velocity takes the name of momentum “p” of the particle, then the equation can be reformulated as “de Broglie’s relation” in the following way:

The wave nature of the electrons was revealed showing that the electron beam can be diffracted. The experiment was made for the first time in 1925 by two American scientists, Clinton Davisson and Lester Germer. They sent a beam of fast electrons against an isolated crystal of nickel. The regular arrangement of the atoms in the crystal acts as a grating, capable of diffracting the waves. Therefore a diffraction image was observed.
George Paget Thomson, in 1927, operating at Aberdeen, Scotland, showed that an electron beam produced a diffraction image passing also through a thin foil of gold, as shown in the figure:

Thus even the electron, like the photon, reveals a double behavior. It is not a quite classical particle, but it can have a detectable wave behavior. The wavelength associated with it is inversely proportional to its momentum.
The “de Broglie’s relation” also applies to the photons:
λ = c/ʋ = hc/hʋ = hc/E = h/(E/c) = h/p
In fact, the momentum of a photon is related to its energy by the relation: p = E/c.
We can conclude by saying that: “all holders of momentum and energy, as electrons, atoms, light, sound and so on, have corpuscular and wave characteristics”.
COMPARISON BETWEEN CLASSICAL MECHANICS AND QUANTUM MECHANICS
Classical Mechanics is a physical theory of deterministic nature. It is governed by the principle of causality: according to the Classical Mechanics, due to the discoveries of Newton and Galileo Galilei, if you know the properties of a body (mass, shape, etc.), its initial conditions of motion (position, velocity, etc.) and the external conditions (force fields, etc.), it is possible to determine, exactly, its behavior instant by instant. Therefore within the framework of the Classical Mechanics the principle of causality applies; namely, in nature nothing happens by chance, each event is determined by an identified cause.
Quantum Mechanics is a physical theory of probabilistic nature. It is based on the concept of probability and observation: for example, if somebody wants accurately to know the location of an electron in an atom, he will never know the speed and vice versa. The famous example of Schrödinger's cat clarifies the nature of Quantum Mechanics:
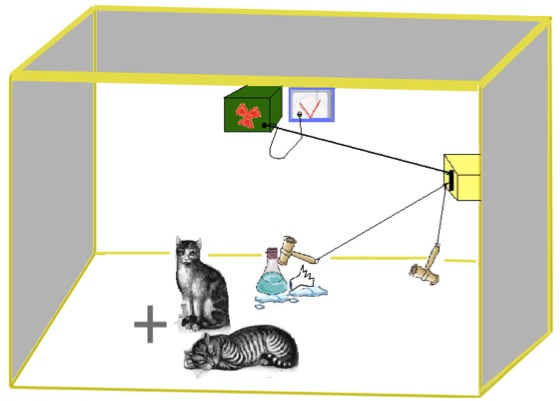
in this example, a cat is closed in a box, with a mechanism connected to a bottle containing poison. The mechanism starts when a radioactive element decays, breaking the bottle containing poison. From the outside, the cat inside the box can be alive or dead, because you don’t know if the radioactive element has decayed or not.
According to the Quantum Mechanics, the cat is in both states “alive and dead”; it is in a superposition of states: “alive” state and “dead” state . Only the observation phase freezes the state of the cat, determining its fate.